EU MDR 2017/745 Essentials Training
Comply Guru offer a European Medical Device Regulation (EU MDR 2017/745) Essentials Course for organizations looking for a concise, high-level introduction for their staff.
Our 2hr Essentials training courses are a highly interactive way to learn at a time & a pace that works best for you, making it easier to get the training you need while balancing a busy home or work life.

Download Brochure
Have 5 or more that need training?
We offer group discounts and are available for in-house (live or virtual) or tailored training in multiple formats.
Our Methodology
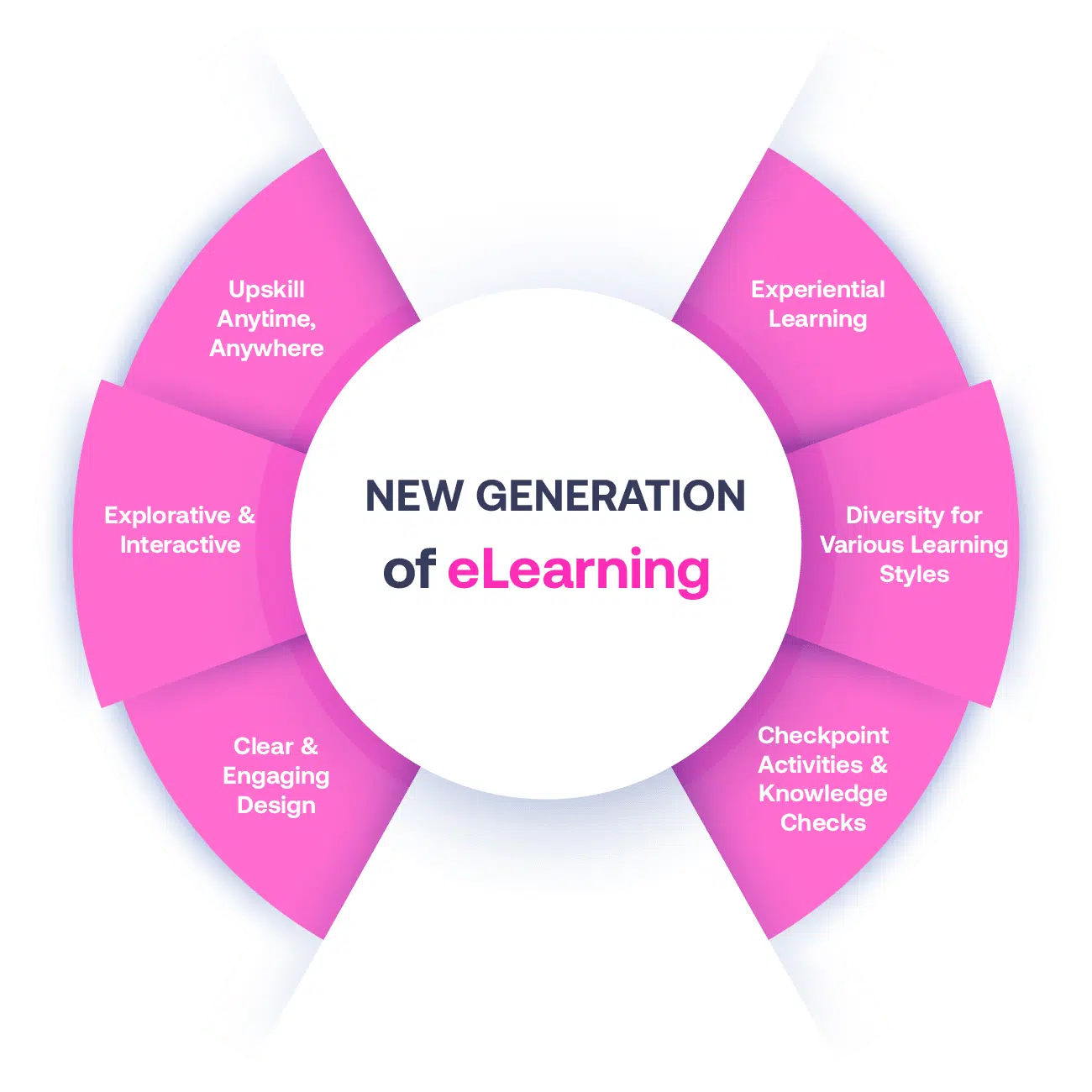
Not all eLearning is the same
Most other providers offer online training that is one-dimensional utilizing either Videos or PowerPoint Presentations. That is not effective training. We deploy a multi-layered methodology that offers you a New Generation of eLearning.
Our Experts
Meet The People Behind The Course
Our experts possess a wealth of industry experience acquired over years of practical application, and in addition, they demonstrate a combination of unwavering passion and a proven aptitude for training.
Course Overview
EU MDR 2017/745 Essentials
Explain the history, purpose, and structure of the EU MDR
Describe the key changes to the EU MDR from the MDD
Identify the different roles and responsibilities associated with the EU MDR
Outline the requirements for placing a medical device on the market
These courses are for organizations who wish to provide their staff with a short, general overview of the EU MDR.
Successful completion will entitle each Learner to receive a digital Certificate of Completion.
In order to successfully complete this course, each Learner will need to:
- Complete all eLearning modules and obtain 70% or higher in the final assessment (MCQ-based)
There are recommended requirements for each Learner in wishing to complete any of our eLearning modules. In our experience, Workplace IT environments’ internal configurations and available software can vary (new or old), and there may be various limitations or other restrictions in place, and as such, the functionality of any Learning Management System (LMS) may be impacted, restricted and may not perform well. Read the full technology requirements here.
Watch and Learn More
About our Essentials Training
Learn about how our MDR Training is leading the industry for innovation through online learning
Customer Reviews
What Our Learners Are Saying
Read verified reviews from Learners who have completed this course.
4.7
Average Rating
186 global ratings
-
it was very good
-
This course was easy to follow and the knowledge checks was a great way to ensure we were paying attention to the course. The information is very relevant.
-
Well done and very informative.
-
I was already well aware of the regulation, so this was a good reminder while bringing some extra info that I was not aware of
-
Excellent introduction to the EU MDR 2017-745 regulation