Regulatory Reliant Internal Auditing for Medical Devices
Regulatory Reliant Internal Auditing for Medical Devices is for existing Internal or Lead Auditors seeking the knowledge & skills to perform internal audits based on integrating ISO 13485, MDSAP and FDA QMSR requirements.
Under Regulatory Reliance models, regulators increasingly depend on the effectiveness of your quality system and the credibility of your internal audit programme to inform audit outcomes, inspection scope, and regulatory confidence.
Many organizations discover this the hard way when internal audits pass, yet MDSAP nonconformities escalate, where FDA inspections uncover gaps that should have been caught internally and when Audit Programmes exist, but do not withstand regulatory scrutiny.
This course is designed for organizations that recognize that internal auditing is now a regulatory control, not a compliance formality. Our live learning methodology focuses on practical ‘learn by doing’ workshops set in a real life context.





Need Corporate, In-House or Customized Training?
Comply Guru offers corporate, in-house and customized training solutions for your organization’s specific needs.
Speak with an experienced member of our team today to learn how we can help.
Course Overview
Regulatory Reliant Internal Auditing for Medical Devices
Conduct integrated internal audits across ISO 13485, MDSAP, and FDA QMSR
Identify and document regulatory critical audit findings
Perform risk-based audit planning and execution
Generate inspection ready audit reports and follow up actions
Operate an internal audit programme aligned with regulatory reliance
Upon successful completion, each Learner shall receive a digital Certificate of Achievement.
Prior to attending this course, learners are expected to have the following prior knowledge:
- A working knowledge of ISO 13485:2016
- An understanding of the MDSAP audit model and requirements
- Working knowledge with the FDA Quality System Regulation (21 CFR Part 820) and/or QMSR
- Prior recognized training in internal or lead auditing (e.g. ISO 13485 Internal Auditor or Lead Auditor course)
- Experience working within a Medical Device Quality Management System (MD-QMS)
To successfully complete this course, each Learner will need to:
- Fully attend the Instructor Workshops as 100% attendance is required.
- Obtain 70% or higher in the graded assessments during the Instructor Workshops
For the live workshops during a virtual delivery, we utilize Microsoft Teams.
Learners need to individually have:
- PC or MAC Computer
- Reliable Internet
- Video Webcam
- Headset or Earbuds
- Quiet Setting
In relation to the eLearning Modules, in our experience, Workplace IT environments’ internal configurations and available software can vary (new or old), and there may be various limitations or other restrictions in place, and as such, the functionality of any Learning Management System (LMS) may be impacted, restricted and may not perform well. Read the full technology requirements here.
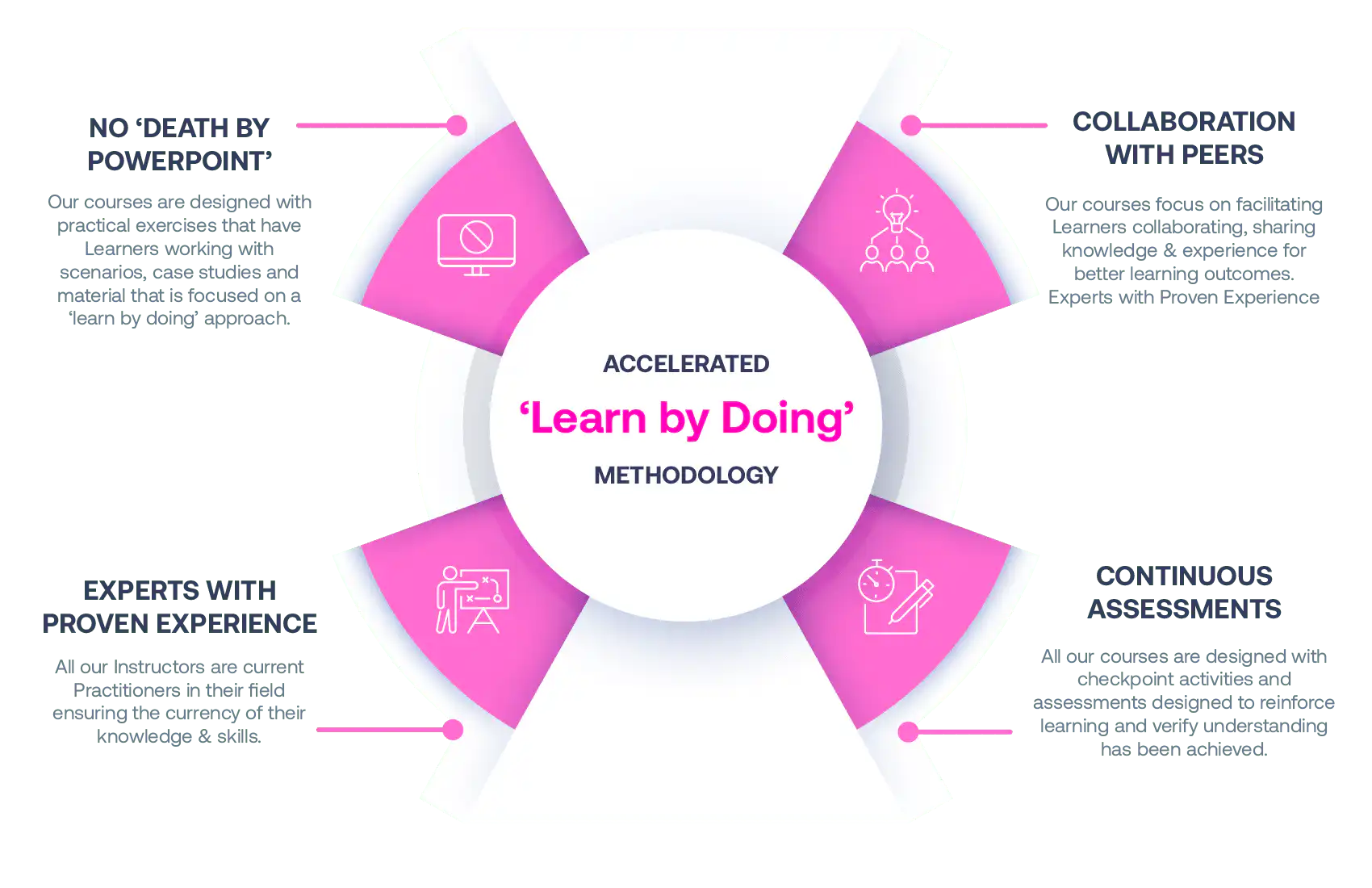
Our Methodology
No More Death By PowerPoint
Our courses are designed with practical exercises that have Learners working with scenarios, case studies and material that is focused on a ‘learn by doing’ approach.
Course Structure Explained
Agenda Overview
Attend live, instructor-led workshops emphasizing practical, real-world application.
| Time | Topic |
|---|---|
| Topics Covered |
|
Our Experts
Meet the Instructors
Our experts possess a wealth of industry experience acquired over years of practical application, and in addition, they demonstrate a combination of unwavering passion and a proven aptitude for training.
Watch and Learn More
About our Internal Auditor Training
Learn about the benefits and key features of Comply Guru’s Instructor led Learning.
Frequently Asked Questions