Clinical Evaluation under EU MDR 2017/745 Training
Clinical Evaluation for Medical Devices Training for those looking to understand the clinical evaluation requirements under the EU MDR (2017/745).
Stay ahead in the medical device industry with our Course on Clinical Evaluation for EU MDR 2017/745 and MEDDEV 2.7/1 Rev. 4 compliance. Designed for regulatory and quality professionals, this course provides a practical understanding of how to meet the requirements for clinical evaluation and reporting under EU MDR.
Through interactive modules and case studies, you’ll learn to navigate clinical data collection, evaluation planning, CER preparation, and Post-Market Clinical Follow-Up (PMCF). This course equips you with the tools and knowledge to facilitate regulatory success.




Need Corporate, In-House or Customized Training?
Comply Guru offers corporate, in-house and customized training solutions for your organization’s specific needs.
Speak with an experienced member of our team today to learn how we can help.
Course Overview
Clinical Evaluation under EU MDR 2017/745
On completion of this course, successful Learners will have the practical knowledge to support their overview of Clinical Evaluation and apply the techniques needed to:
- Define the requirements for clinical evaluation under EU MDR 2017/745.
- Explain the role of MEDDEV 2.7/1 Rev. 4 guidance in Clinical Evaluation
- Differentiate between clinical data sources and their applicability
- Develop and Document a compliant Clinical Evaluation Report (CER)
- Clinical Evaluation and Post-Market Clinical Follow-up (PMCF)
Prior to attending this course, learners are expected to have the following prior knowledge:
EU MDR 2017/745
- Familiarity with the structure and key provisions of the EU Medical Device Regulation 2017/745.
- Basic knowledge of medical device classification and conformity assessment procedures under EU MDR.
MEDDEV 2.7/1 Rev. 4
- Familiarity with MEDDEV 2.7/4 Rev. 4
- Quality Professionals
- Quality Engineers
- Regulatory Professionals
Successful completion will entitle each Learner to receive a digital Certificate of Completion within 1 business day.
In order to successfully complete this course, each Learner will need to:
- Fully attend the Live Instructor Workshop as 100% attendance is required
If you are completing this training virtually, the following applies:
For the live workshops during a virtual delivery, we utilize both Zoom and Microsoft Teams.
Learners need to individually have:
- PC or MAC Computer
- Reliable Internet
- Video Webcam
- Headset or Earbuds
- Quiet Setting
In relation to the eLearning Modules, in our experience, Workplace IT environments’ internal configurations and available software can vary (new or old), and there may be various limitations or other restrictions in place, and as such, the functionality of any Learning Management System (LMS) may be impacted, restricted and may not perform well. Read the full technology requirements here.
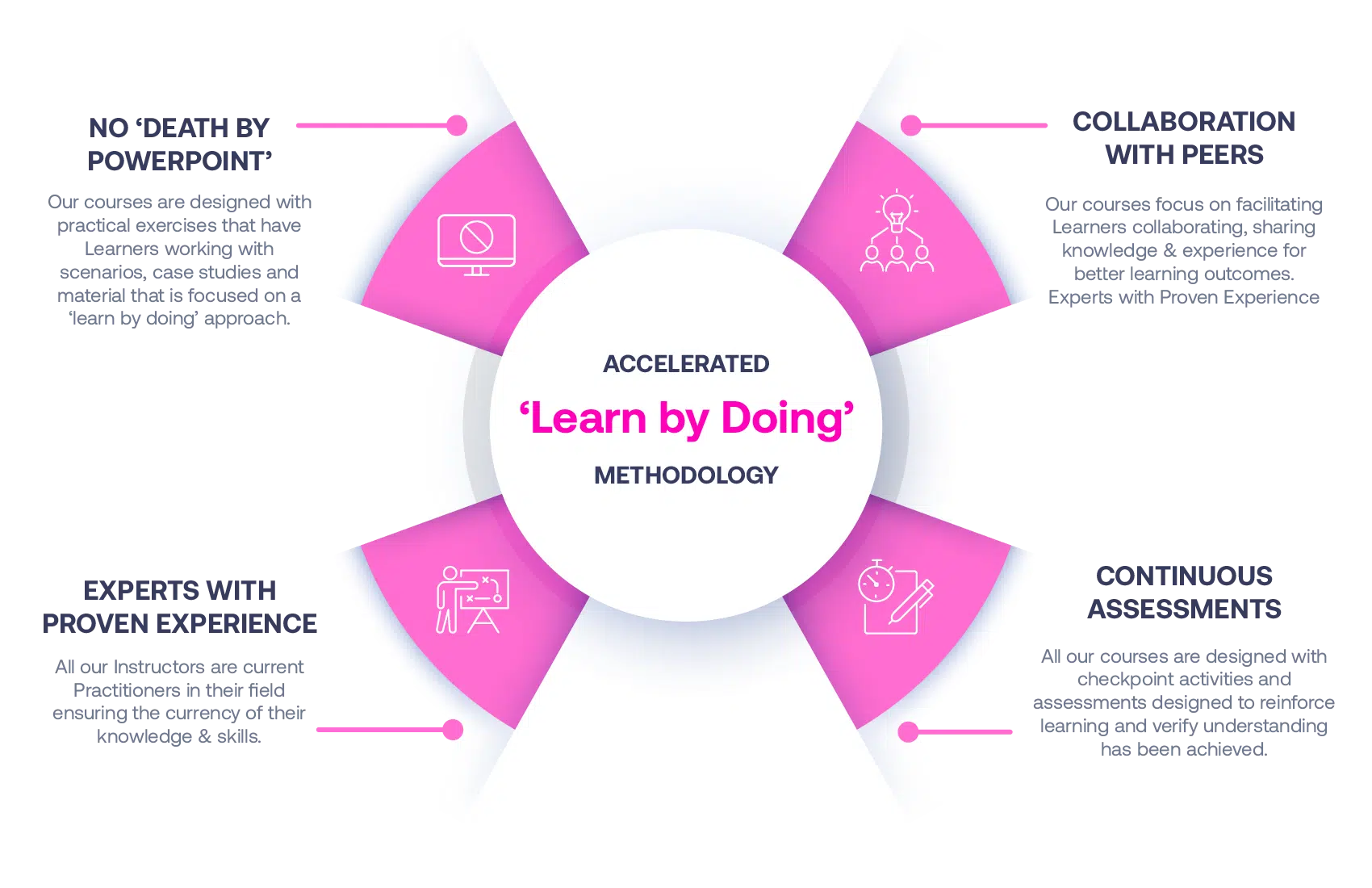
Our Methodology
No More Death By PowerPoint
Our courses are designed with practical exercises that have Learners working with scenarios, case studies and material that is focused on a ‘learn by doing’ approach.
Course Structure Explained
Detailed Breakdown & Agenda
Attend live, instructor-led workshops emphasizing practical, real-world application.
| Time | Topic |
|---|---|
| #1 | Introduction to Clinical Evaluation
Practical Workshop Identify Clinical Evaluation Requirements for a Medical Device |
| #2 | Regulatory Requirements for Clinical Evaluation
Practical Workshop Assessing Compliance with EU MDR Clinical Evaluation Requirements |
| #3 | Sources of Clinical Data
Practical Workshop Evaluating the Adequacy of Clinical Data |
| #4 | Preparing a Clinical Evaluation Plan (CEP)
Practical Workshop Development/Review of a PMS Plan |
| #5 | Writing a Clinical Evaluation Report (CER)
Practical Workshop Development/Review of a CER |
| #6 | Post-Market Clinical Follow-up (PMCF)
Practical Workshop Designing a PMCF Plan |
Our Experts
Meet The Instructors
Our experts possess a wealth of industry experience acquired over years of practical application, and in addition, they demonstrate a combination of unwavering passion and a proven aptitude for training.
Watch and Learn More
About our Clinical Evaluation for Medical Devices Training
Learn about the benefits and key features of Comply Guru’s Instructor led Learning.
Customer Reviews
What Our Learners Are Saying
Read verified reviews from Learners who have completed this course.
4.8
Average Rating
4 global ratings
-
Excellent training. A lot covered over 1 day.
-
Trainer was very knowledgeable and open to answering questions throughout the training. the trainer gave very good information to support the requirements of Clinical evaluation Reports.
Frequently Asked Questions