Webinars
Show Me All Webinars
- eu mdr
- medical devices
- internal auditing
- unannounced audits
- quality
- regulatory affairs
- technical documentation
- medtech
- open day
- qara
- training
- fda
- final rule
- manufacturers
- qmsr
- auditing
- mdsap
- version 9
- post market surveillance
- capa
- fda qmsr
- iso 13485
- gap analysis
- transition

Navigating the FDA QMSR Transition: A Practical Gap Analysis Approach
This is a Free Comply Guru Webinar where Navigating the FDA QMSR Transition: A Practical Gap-Analysis Approach will be discussed by Michelle Keane.

How to Prevent Weak CAPA Investigations
This is a Free Comply Guru Webinar where How to Prevent Weak CAPA Investigations will be discussed by Michelle Keane and special guest Jackie Torfin.

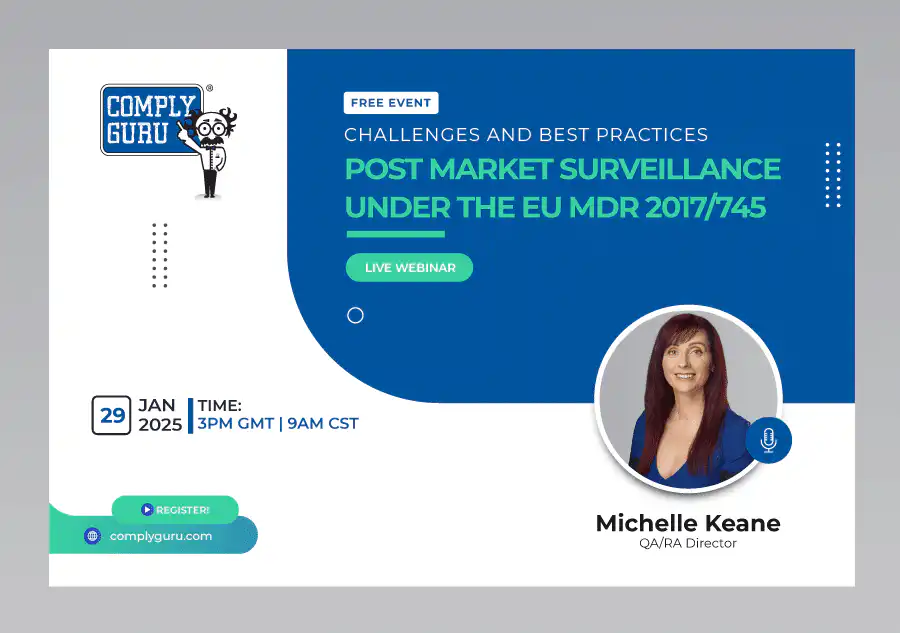
Post Market Surveillance under the EU MDR – Challenges and Best Practices
This is a Free Comply Guru Webinar where Post-Market Surveillance under the EU MDR – Challenges and Best Practices will be discussed by Michelle Keane.

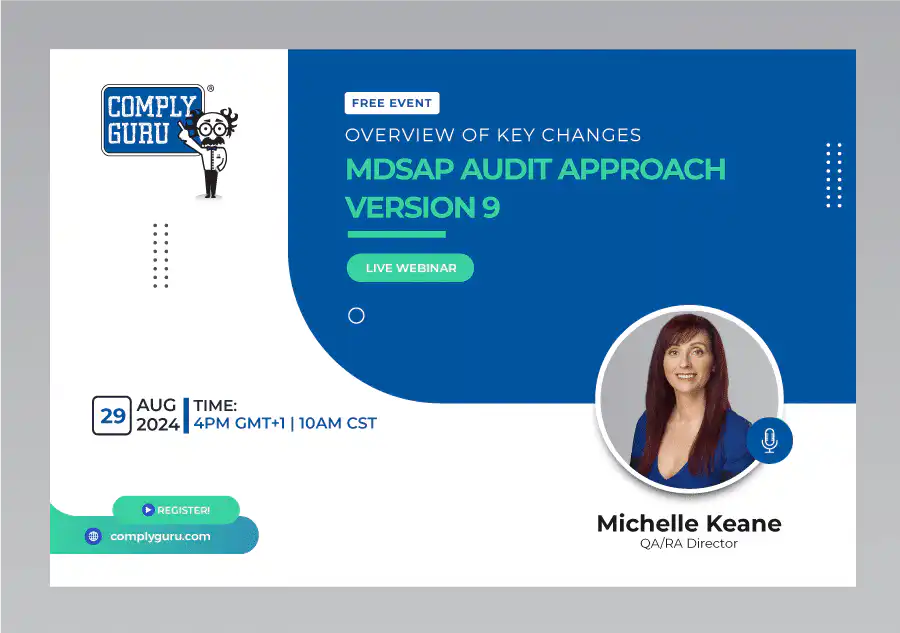
Overview of Key Changes with MDSAP Audit Approach Version 9
This is a Free Comply Guru Webinar covering an Overview of Key Changes with MDSAP Audit Approach Version 9 with Michelle Keane.

Impact of the FDA QMSR on Medical Device Manufacturers
This is a Free Comply Guru Webinar where the new FDA QMSR Final Rule and its impact on Manufacturers will be discussed by Michelle Keane.

MedTech Quality and Regulatory Affairs (QARA) Training Open Q&A
Planning ahead on training needs? Looking to budget? Join us on Wednesday, November 6th 2024, from 4pm GMT, where Comply Guru’s Eoin P. Kelly & Michelle Keane will discuss Comply Guru’s MedTech Quality and Regulatory Affairs Training Portfolio. It is a great opportunity to come and ask questions about any course!

Technical Documentation under the EU MDR 2017/745
This is a Free Comply Guru Webinar where Technical Documentation under the EU MDR will be discussed by Michelle Keane and Orla Keane.

Unannounced Audits in the Medical Device Industry
This is a Free Comply Guru Webinar on Unannounced Audits with Michelle Keane.

Four Considerations when Internal Auditing under the EU MDR Webinar
This is a Free Comply Guru Webinar on Key Considerations when Internal Auditing under the EU MDR with Michelle Keane.

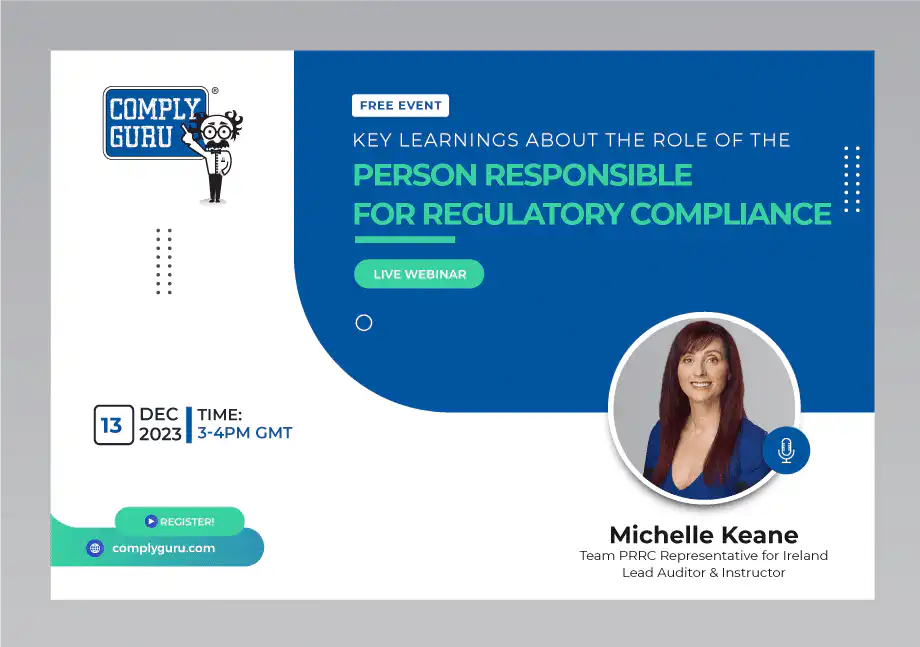
The Role of the Person Responsible for Regulatory Compliance (PRRC) Webinar
This is a Free Comply Guru Webinar on the Role of the Person Responsible for Regulatory Compliance (PRRC) under the EU MDR and EU IVDR with Michelle Keane.